Hi! I’m Laura.
I’m a Postdoctoral Neuroscientist at the Krembil Brain Institute & University Health Network in Toronto, Canada. I currently work in the Bains Lab where I use computational neuroscience techniques and advanced data analytics to understand the role of touch in stress.
I am supported by a CIHR Canada Postdoctoral Research Award (2026-Present) and a Canada Leads Scholarship (2026-Present) and was previously funding by a CIHR Doctoral Graduate Scholarship (2022–2025) and SickKids Research Training Competition (RTC) Scholarship (2020–2022).
View my CV/resumé. (Last Updated: April 2026)
- Computational Neuroscience
- Neural Coding
- Somatosensation
- Pain & Stress
PhD in Biomedical Engineering, 2025
University of Toronto, Canada
MSc in Neuroscience, 2018
Queen's University, Canada
BSc in Life Sciences/Mathematics, 2016
Queen's University, Canada
Featured Research
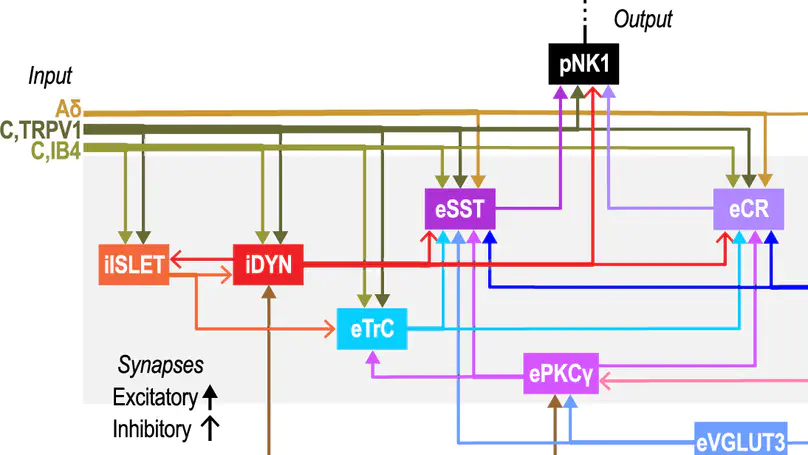
Building a model of the spinal dorsal horn to investigate chronic pain.
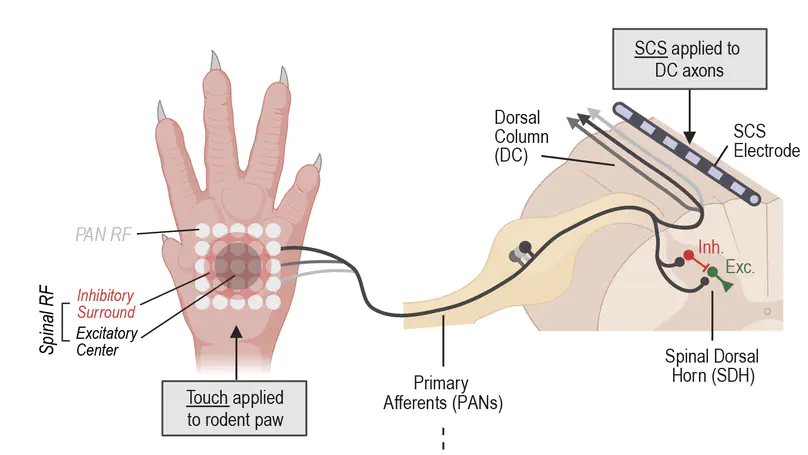
Weakening of synaptic inhibition in the spinal dorsal horn (SDH) contributes to mechanical allodynia after peripheral nerve pathology. Restoring inhibition can alleviate allodynia, whereas weakening it is sufficient to induce allodynia and spontaneous pain in uninjured conditions. Disinhibition is known to un-gate nociceptive polysynaptic spinal circuits, but why allodynia is predominantly evoked by certain touch stimuli remains unclear. To address this, we incorporated receptive fields (RFs) into a computational model of the SDH to study the processing of stimuli with different spatiotemporal features. Our model reveals that broad stimuli normally suppress spinal output by engaging inhibition from the RF’s inhibitory surround, but previously subliminal excitation can be engaged when inhibition is compromised, fundamentally altering excitatory–inhibitory balance. The efficacy of spinal inhibition also depends on the input’s temporal pattern, especially since excitatory and inhibitory spinal neurons are preferentially sensitive to synchronous and asynchronous input, respectively. Furthermore, spikes driven by synchronous input are resistant to feedforward inhibition. This combination of effects may explain why broad dynamic touch (e.g., brush) evokes more allodynia than punctate static touch. On the other hand, asynchronous and spatially disordered input like that evoked by kilohertz-frequency spinal cord stimulation was found to preferentially activate inhibitory neurons, thus reducing allodynia. Overall, our results suggest how spatial and temporal stimulus features impact the flow of sensory input through disinhibited spinal circuits. Our results show how quantitative computational models can connect injury-induced molecular changes to clinically relevant sensory effects by revealing nonintuitive processes occurring at the cellular and circuit levels.
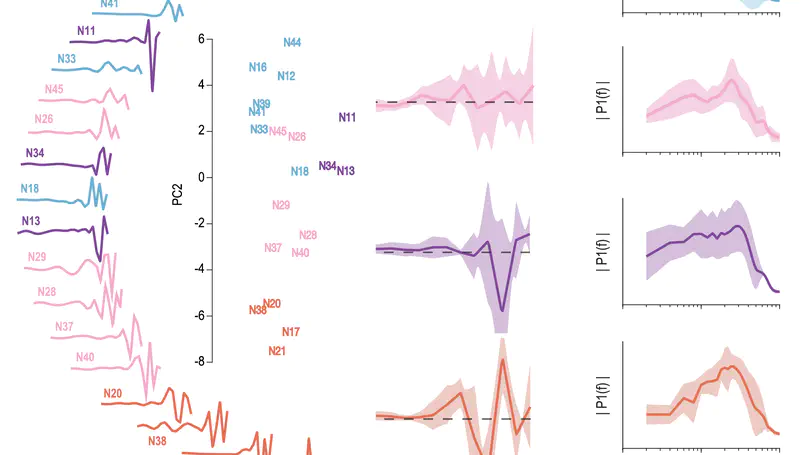
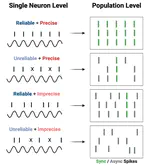
Our sense of touch starts with activation of nerve fibres in the skin. Although response properties of various fibre types are well-established in other species (e.g. primates), quantitative characterization in rats and mice is limited. To fill this gap, we performed a comprehensive electrophysiological investigation into the coding properties of tactile fibres in rodent non-hairy skin and then simulated these fibres to explain differences in their responses. We show that rodent tactile fibres resemble those from other species, but that their heterogeneity at the population level may differ, with potentially important implications for encoding of touch. Simulations reveal intrinsic mechanisms that support this heterogeneity and provide a useful tool to explore somatosensation in rodents.
Publications
Selected Presentations
From seminars & conferences